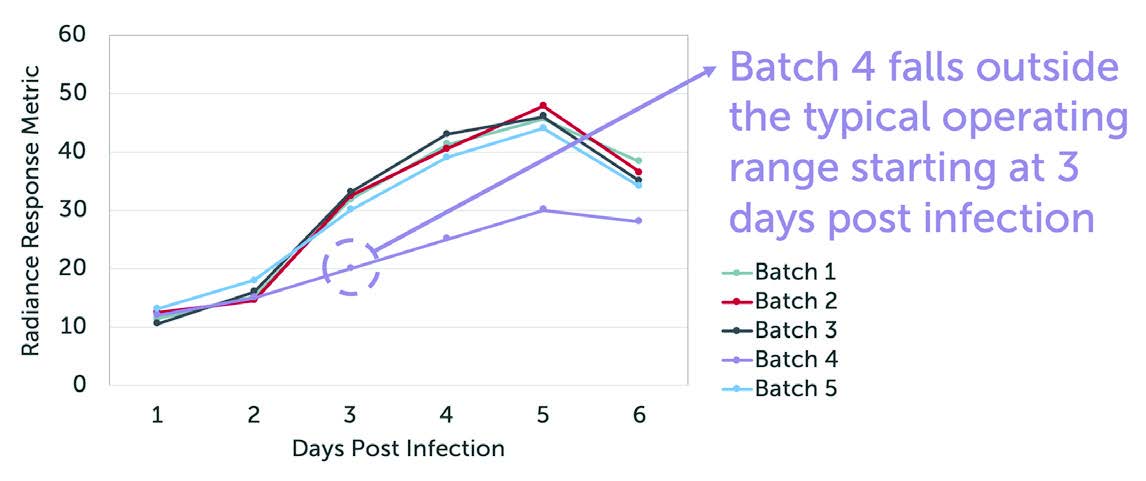
Quantify Detectable Differences.
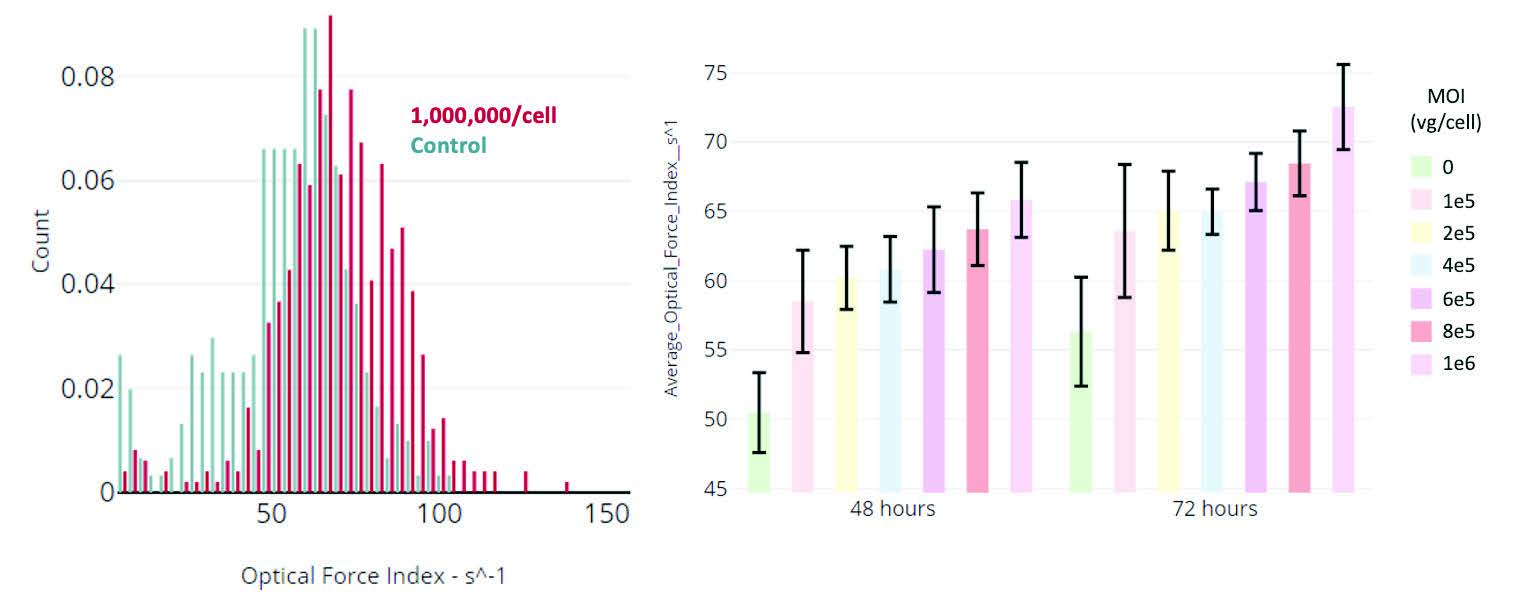
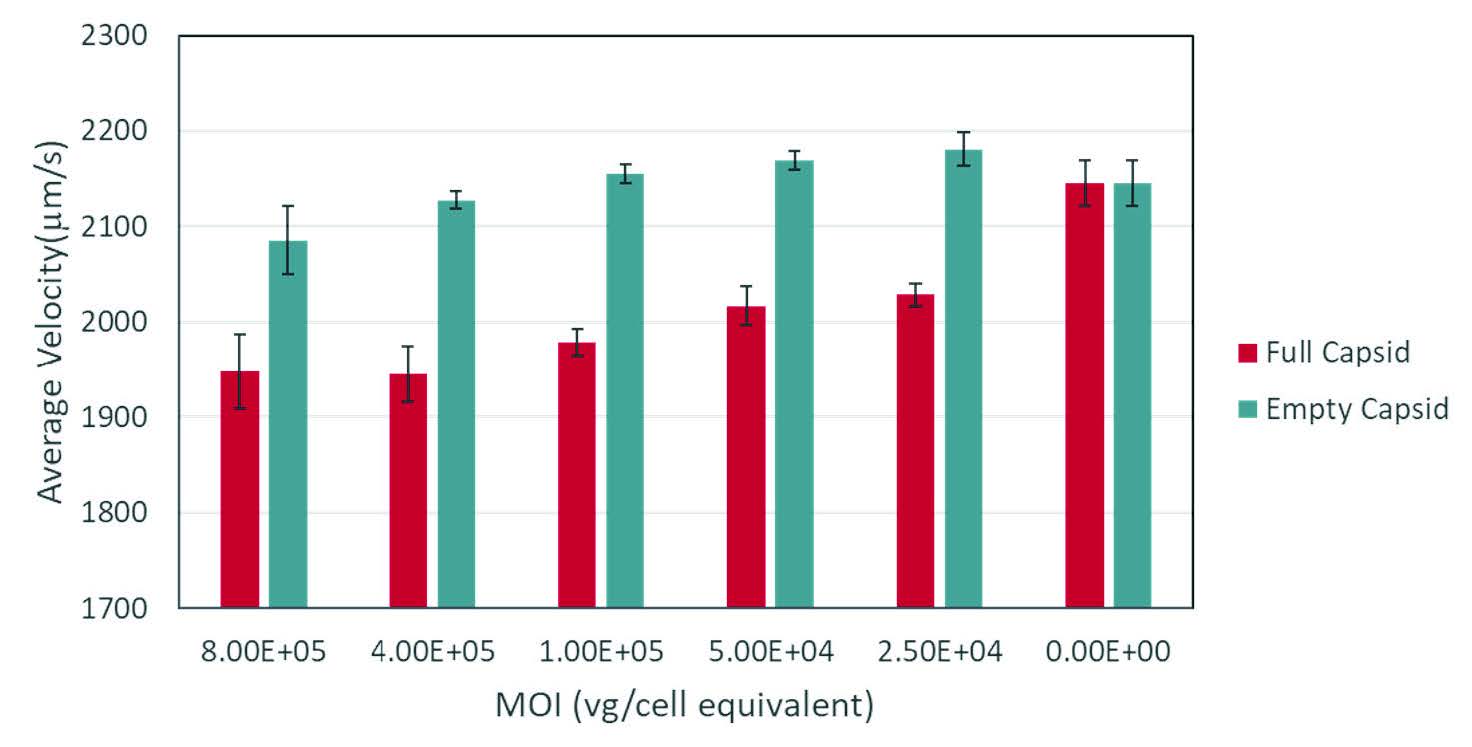
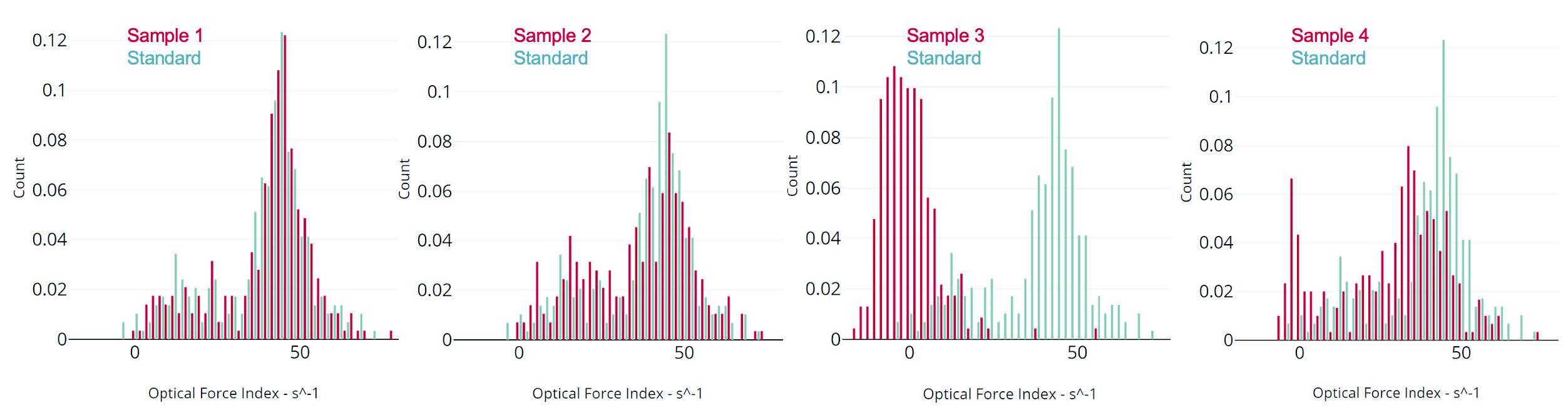
Differences in cell biochemistry, morphology, and deformability (cytoskeletal changes), which are often associated with viral infection, cancer, sepsis, and other diseases, give rise to detectable differences in optical force and deformability which are quantitated by Radiance.
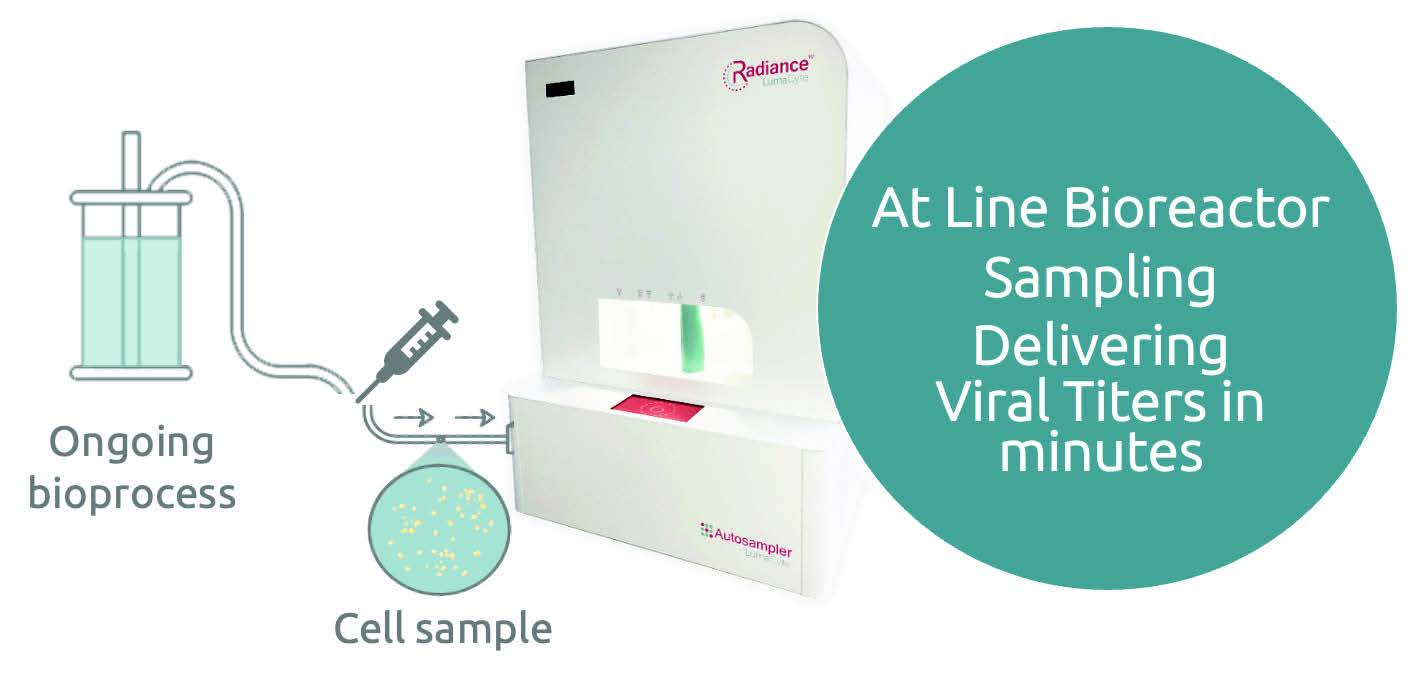
Fits within your current workflow.
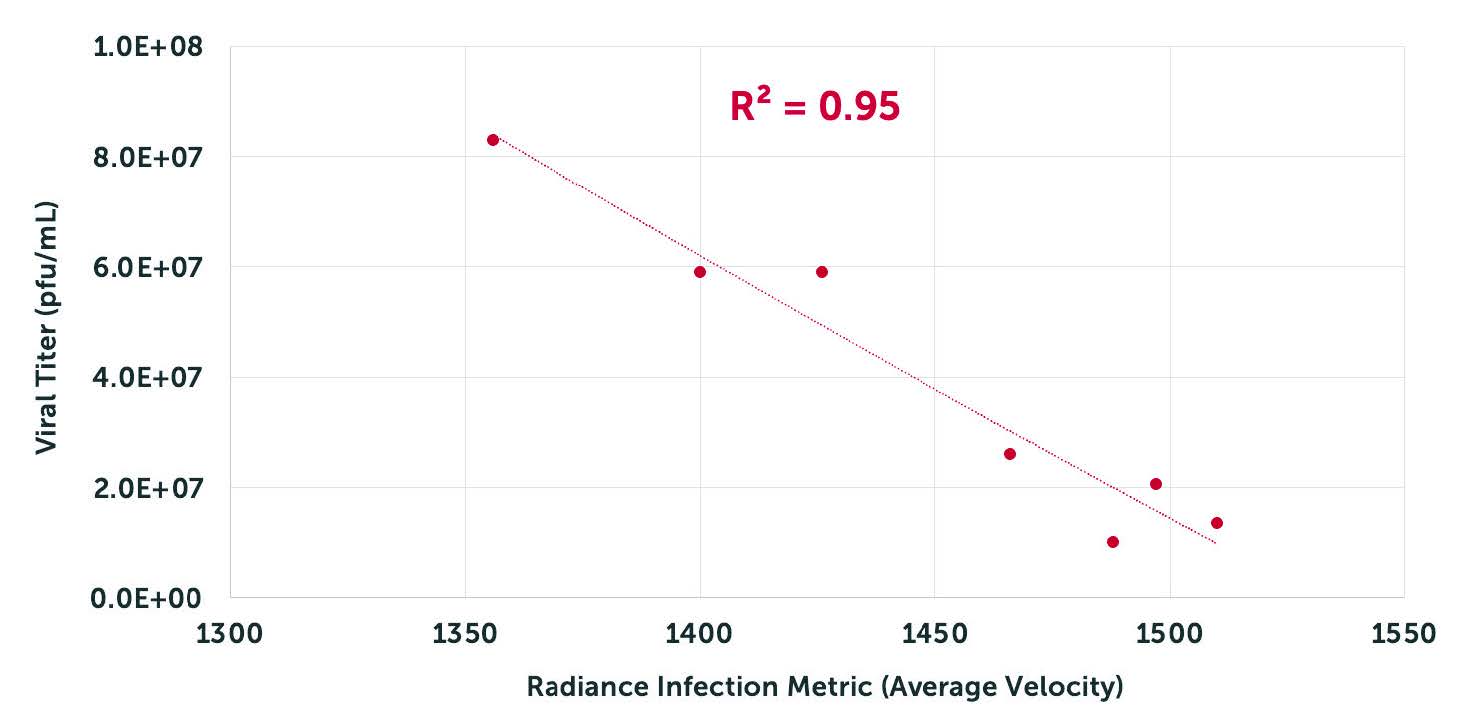
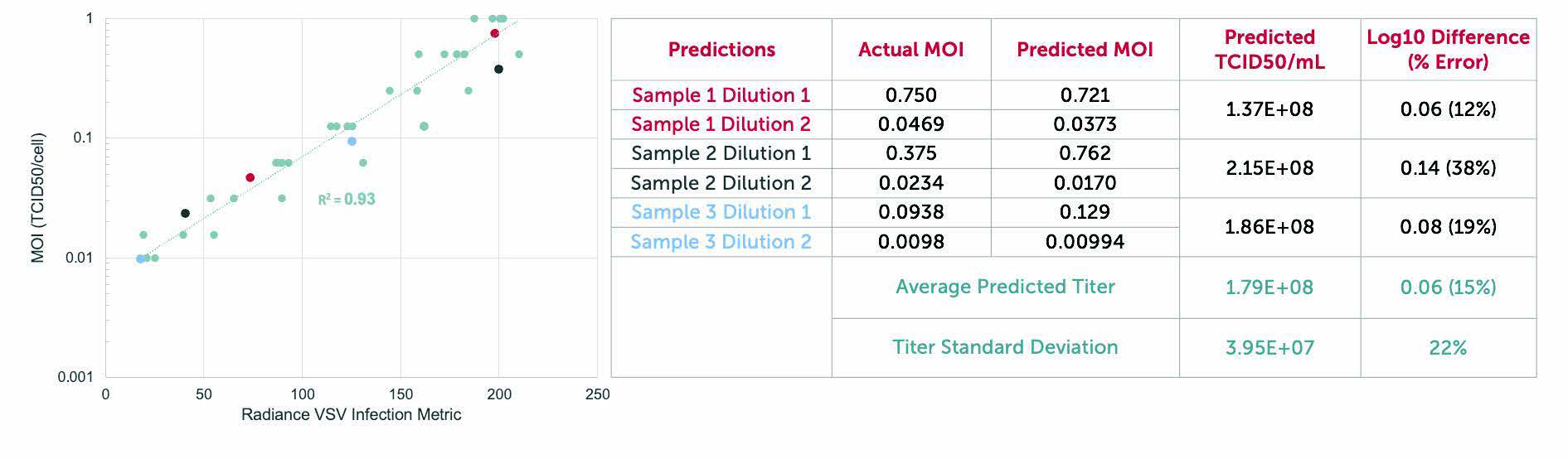
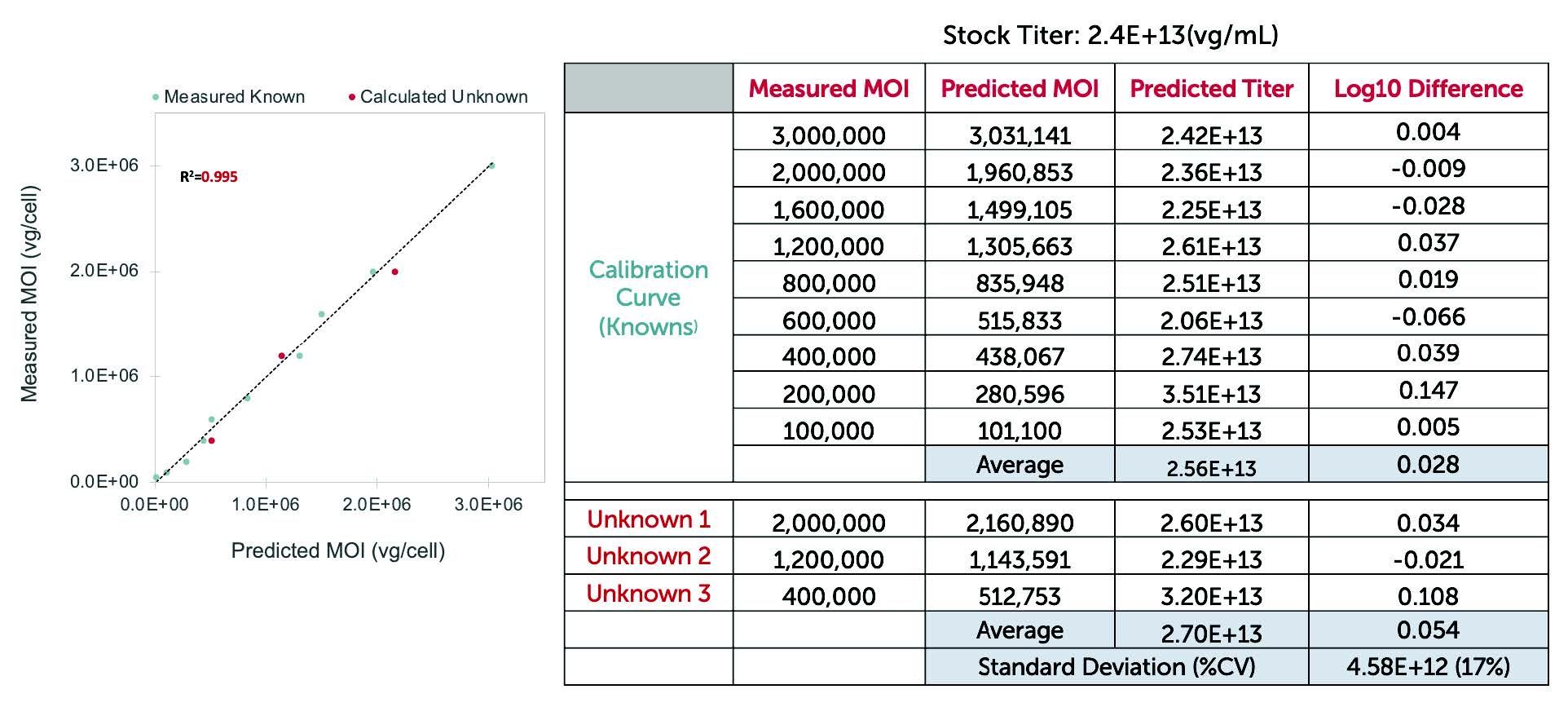
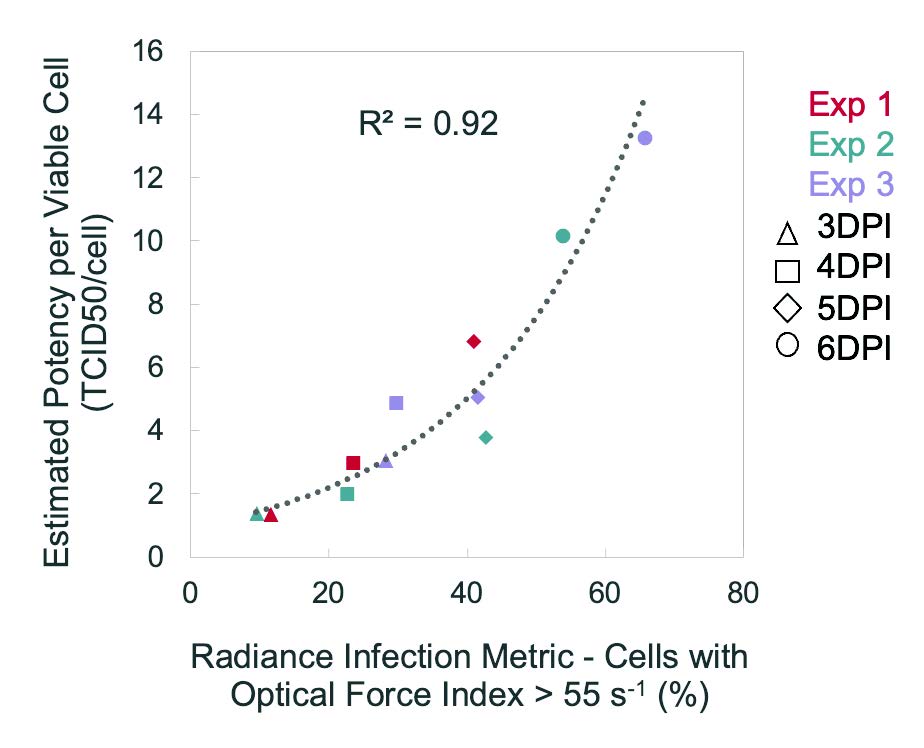
Correlates tightly with ddPCR, TCID50, and Plaque Assays.